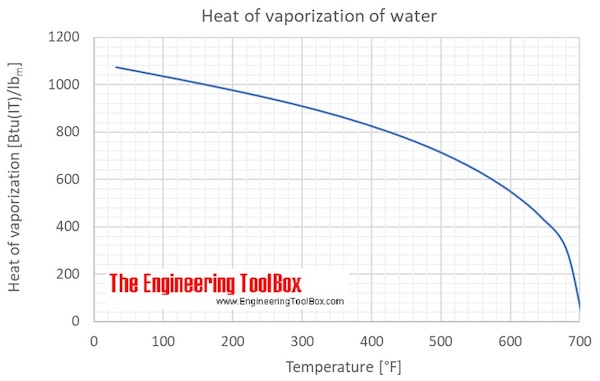
Definition of heat of vaporization. The heat of fusion of ice is 333 Jg meaning 333 J is absorbed when 1 gram of ice melts The heat of vaporization of liquid water at 100C is 2257 Jg.
Latent Heat Definition Types Formula Fusion And Vaporization
The latent heat of vaporization for water is 226 x 105 Jkg.
Heat of vaporization definition for dummies. Due to the increases in kinetic energy the force of attraction between the molecules reduces. This means that 226 x 105 J of heat energy must be added to turn one kilogram of water from liquid to gas at 100 degrees Celsius. Distillation is one of the most practical methods for separation and purification of chemical compounds.
Heat of vaporization The amount of heat required to convert a liquid into a gas at constant temperature and pressure. The heat of fusion also known as the latent heat of fusion is a category of latent heat describing the energy for the phase change between a liquid and a solid to occur without a change in. The enthalpy of vaporization also known as the heat of vaporization or heat of evaporation is the amount of energy that must be added to a liquid substance to transform a quantity of that substance into a gas.
As a result of an increase in temperature the kinetic energy of the molecules increases. Heat of vaporization - heat absorbed by a unit mass of a material at its boiling point in order to convert the material into a gas at the same temperature heat of vaporisation heat of transformation latent heat - heat absorbed or radiated during a change of phase at a constant temperature and pressure. The amount of heat required to evaporate a liquid System and its Surroundings.
The enthalpy of vaporization is a function of the pressure at which that transformation takes place. A gas releases the same amount of heat when it. The heat applied to effect a change of state at the boiling point is the latent heat of vaporization.
The enthalpy of vaporization is a function of the pressure at which that transformation takes place. Enthalpy is a concept used in science and engineering when heat and work need to be calculated. You can use the heat of fusion of ice and heat of vaporization of water to calculate the enthalpy change when ice melts into a liquid and the liquid turns to a vapor.
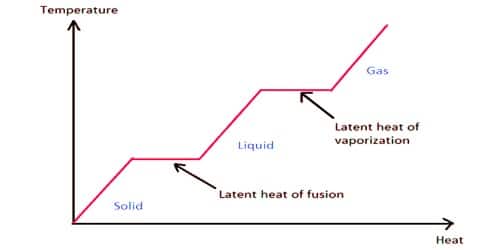
The concentration of a gas is given by its vapor pressure. Latent comes from the Latin latere which means to lie hidden or concealed. The amount of heat required to convert 1 g of ice to 1 g of water 80 Cal is termed the latent heat of melting and it is higher for water than for any other commonly occurring substance.
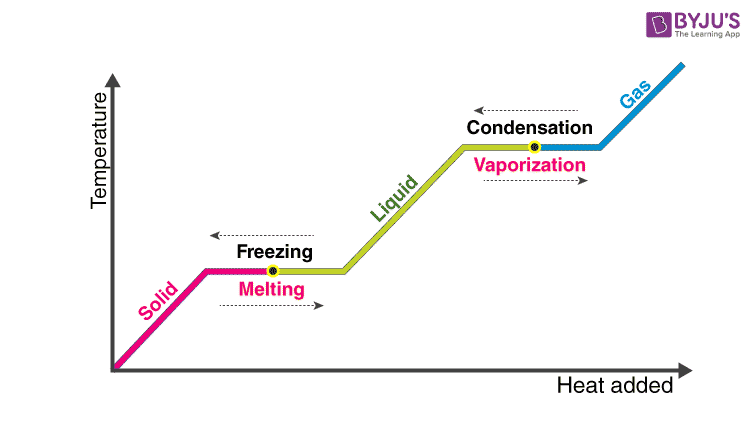
The enthalpy of vaporization is often quoted for the normal boiling temperature of the substance. Heat of Vaporization. Latent heat is the additional heat required to change the state of a substance from solid to liquid at its melting point or from liquid to gas at its boiling point after the temperature of the substance has reached either of these points.
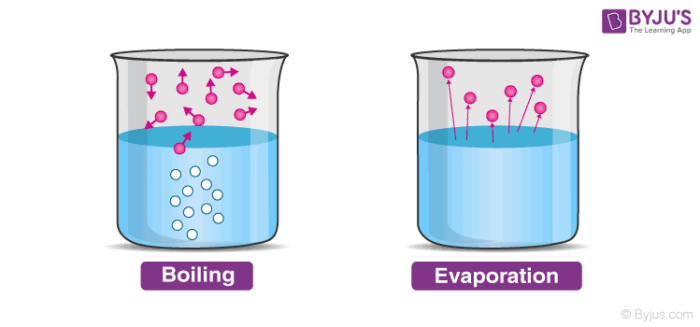
Vaporization can be defined as the process in which liquid state changes into the vapour state. The system is the area in which a reaction takes place and the surroundings is the area that interacts with the system. The name comes from the Greek word enthalpos meaning to put heat into.
The idea and the word were made up by the Dutch scientist Heike Kamerlingh Onnes in 1909. The latent heat of vaporization H vap also known as the enthalpy of vaporization or evaporation is the amount of energy enthalpy that must be added to a liquid substance to transform a given quantity of the substance into a gas. Heat absorbed when a liquid vaporizes specifically.
The heat of vaporization is the heat required to completely vaporize a unit of saturated liquid or condense a unit mass of saturated vapor and it equal to hlg hg hl. The heat of vaporization is a latent heat. The heat of vaporization is the fundamental quantity that determines the experimental conditions at which an industrial or laboratory-scale distillation should be run.
The quantity of heat required at a specified temperature to convert unit mass of liquid into vapor.
Learn About Latent Heat Of Vaporization Chegg Com
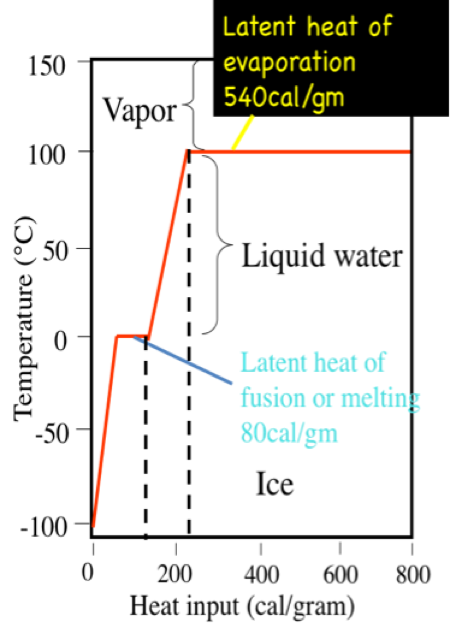
Latent Heat And Freezing And Boiling Points Earth 111 Water Science And Society
Science Universe Physics Articles Latent Heat Of Vaporization And Of Fusion Explained By The Kinetic Theory

Heat Of Vaporization Experiment
What Is The Practical Use Of Latent Heat Of Evaporation Quora
Science Universe Physics Articles Latent Heat Of Vaporization And Of Fusion Explained By The Kinetic Theory

Heat Of Fusion Heat Of Vaporization Concept Chemistry Video By Brightstorm

A Cartoon Guide To Physics Latent Heat Of Vaporization Latent Heat Physics Classroom Heat Transfer Science

Enthalpy Of Vaporization Of Hydrazine Monomethylhydrazine And Download Scientific Diagram

Latent Heat Of Fusion And Vaporization Specific Heat Capacity Calorimetry Physics Youtube
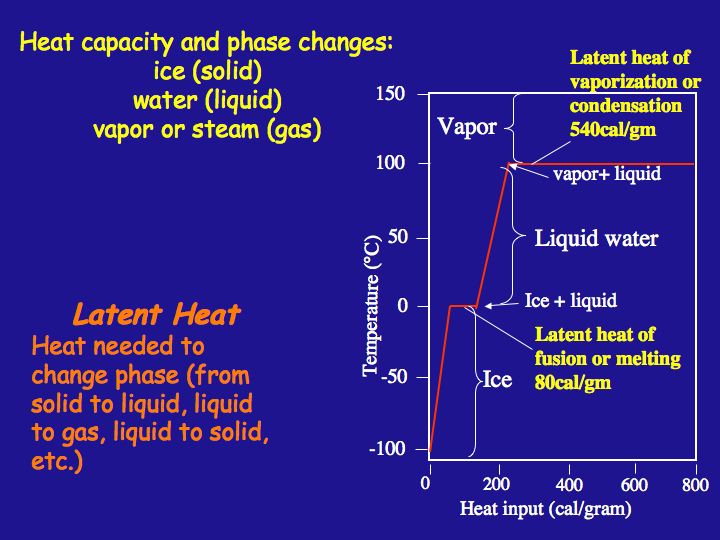
Latent Heat Heat Capacity Earth 540 Essentials Of Oceanography For Educators

Differentiate Latent Heat Of Fusion And Latent Heat Of Vaporization Qs Study

Caesium Specific Heat Latent Heat Of Fusion Latent Heat Of Vaporization Nuclear Power

Heat Of Vaporization An Overview Sciencedirect Topics

Difference Between Latent Heat Of Fusion And Vaporization Compare The Difference Between Similar Terms