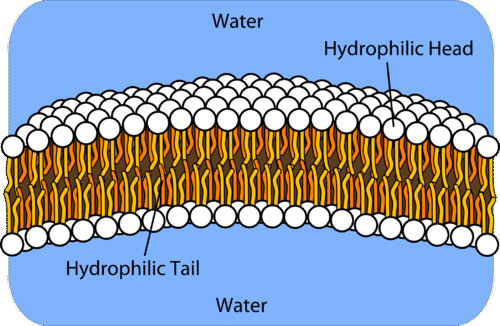
Phospholipids are a major component of cell membranes which enclose the cytoplasm and other contents of a cell. The resulting structure is called a.
How Do Phospholipids Interact With Water Molecules Quora
The nonpolar tails attract water because water is polar and opposites attract.

How do phospholipids interact with water. The hydrophilic regions of the phospholipids tend to form hydrogen bonds with water and other polar molecules on both the exterior and interior of the cell. The nonpolar tails attract water because water is. Thus the membrane surfaces that face the interior and exterior of the cell are hydrophilic.
Term for having both a hydrophilic and hydrophobic region. Phospholipids do not interact with water because water is polar and lipids are nonpolar. In the PLB the phosphate containing head group eg.
In fact they are an essential component of the cell membrane. 3 qualities of saturated fatty acids. The nonpolar tails do not.
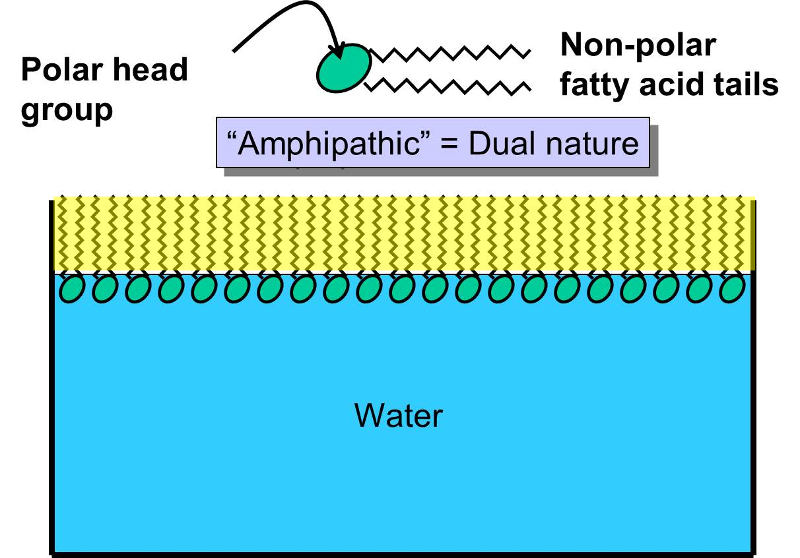
1 Phospholipids do not interact with water because water is polar and lipids are nonpolar. This means that they have a hydrophilic polar phosphate head and two hydrophobic fatty acid tails. When phospholipids are mixed with water they spontaneously rearrange themselves to form the lowest free-energy configuration.
The hydrophobic tails associate with one another forming the. 2 The polar heads avoid water. Because of this polarnon-polar interaction with water or the aqueous protein solution it makes the formation of.
Macromolecule phophate containing head glycerol backbone 2 fatty acid tails amphipathic. The non-polar tails do not. The head is polar.
This means that the hydrophobic regions find ways to remove themselves from water while the hydrophilic regions interact with water. This means that the hydrophobic regions find ways to remove themselves from water while the hydrophilic regions interact with water. When phospholipids are mixed with water they spontaneously rearrange themselves to form the lowest free-energy configuration.
The way that phospholipids interact with water is very important in. In other words they act like a fat or an oil avoiding water. Phospholipids are one of the main types of molecules in the body.
In aqueous solutions phospholipids are driven by hydrophobic interactions that result in the fatty acid tails aggregating to minimize interactions with water molecules. The polar heads interact with water. Phosphatidylcholine points towards the aqueous outside of both sides of the bilayer and the hydrophobic fatty acid tail groups point towards each other building the inside of the bilayer thus.
The polar heads avoid water. These components of the phospholipids cause them to orientate themselves so the phosphate head can interact with water and the fatty acid tails cant hence forming a bilayerDec 5 2017. 2 The polar heads avoid water.
The polar head region faces outward and interacts with the liquid. The phospholipid bilayer consists of two adjacent sheets of phospholipids arranged tail to tail. The nonpolar tails do not-They have hydrophilic tails that face outward and are exposed to water and hydrophobic heads that face the center of the membrane and are shielded from water-The polar heads repel water and the nonpolar tails attract water.
How do membrane phospholipids interact with water-The polar heads interact with water. The phospholipids are amphiphilic. Because of this polarnon-polar interaction with water or the aqueous protein solution it makes the formation of phospholipid bilayers PLBs possible at all.
When placed in water phospholipids will orient themselves into a bilayer in which the non-polar tail region faces the inner area of the bilayer. The hydrophilic end usually contains a negatively charged phosphate group and the hydrophobic end usually consists of two tails that are long fatty acid residues. Phospholipids link side by side forming what.
In contrast the interior of the cell membrane is hydrophobic and will not interact with water. 5 qualities of a phospholipid. As the phosphate groups are polar and hydrophilic they are attracted to water in the intracellular fluid.
3 Phospholipids dissolve in water. How do phospholipids interact with water molecules. The polar heads interact with water.
The resulting structure is called a. Orrect answer orrect answer Phospholipids dissolve in. The nonpolar tails attract water because water is polar and opposites attract.
Since the heads face outward one layer is exposed to the interior of the cell and one layer is exposed to the exterior. Phospholipids dissolve in water. Saturated and unsaturated fatty acids.
The fatty acid tails are non-polar making them hydrophobic water fearing. In terms of how phospholipids interact with water they have have a split personality. Phospholipids do not interact with water because water is polar and lipids are nonpolar.
1 Phospholipids do not interact with water because water is polar and lipids are nonpolar. Phospholipids do not interact with water because water is polar and lipids are non-polar. The hydrophilic polar head group interacts with water by being attracted to it and the hydrophobic non-polar tail by being pushed away from water.

The Lift Gymsupplementing Fats With More Fats The Lift Gym

Phospholipids Biology For Majors I
With Embedded Q A Lec 4 Biol C2005 F2401 L Chasin Sept 16 2010 Last Updated Thursday September 16 2010 12 21 Am C Copyright 2010 Lawrence Chasin And Deborah Mowshowitz Department Of Biological Sciences Columbia University New
How Do Phospholipids Interact With Water Molecules Quora

Phospholipid Structure Function What Is A Phospholipid Video Lesson Transcript Study Com

Structural Biochemistry Lipids Lipid Bilayer Wikibooks Open Books For An Open World
Http Www Idc Online Com Technical References Pdfs Chemical Engineering Intermolecular Forces In Biology Lipids And Lipid Aggregates Pdf

What Function Does A Phospholipid Serve In A Cell Membrane Quora

Structure Of The Membrane Biology For Majors I
Since A Lipid Bilayer Has Both Hydrophobic And Hydrophilic Ends How Is It Possible That A Lipid Soluble Substance Can Pass The Membrane Quora
26 9 Phospholipids Chemistry Libretexts

Problem When Phospholipids Are Mixed With Water Their

Water Cell Membrane Interactions
Overview Of Cell Structure And Function
Introduction And Cell Membrane
26 9 Phospholipids Chemistry Libretexts